2024
Stanford researchers make critical COVID-19 discovery
After 5 years with COVID-19, health experts say there is still much to learn about the virus that causes the disease. At the height of the pandemic, hospitals were overwhelmed with patients with severe cases of the respiratory virus including life-threatening complications. It was believed that a certain type of lung cell made some people more susceptible to severe infection but now that is no longer the case. KTVU's Heather Holmes speaks with Stanford scientists Catherine Blish and Mark Krasnow about their critical new research that found a different suspected source of vulnerability.
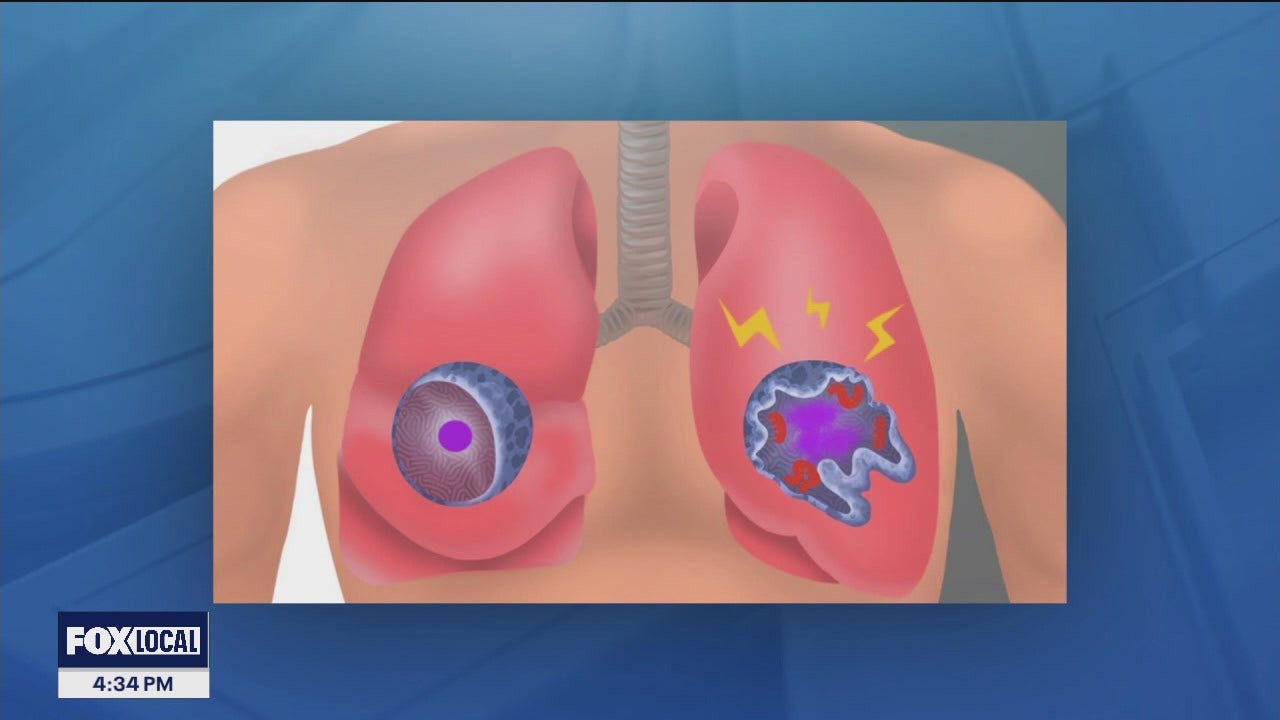
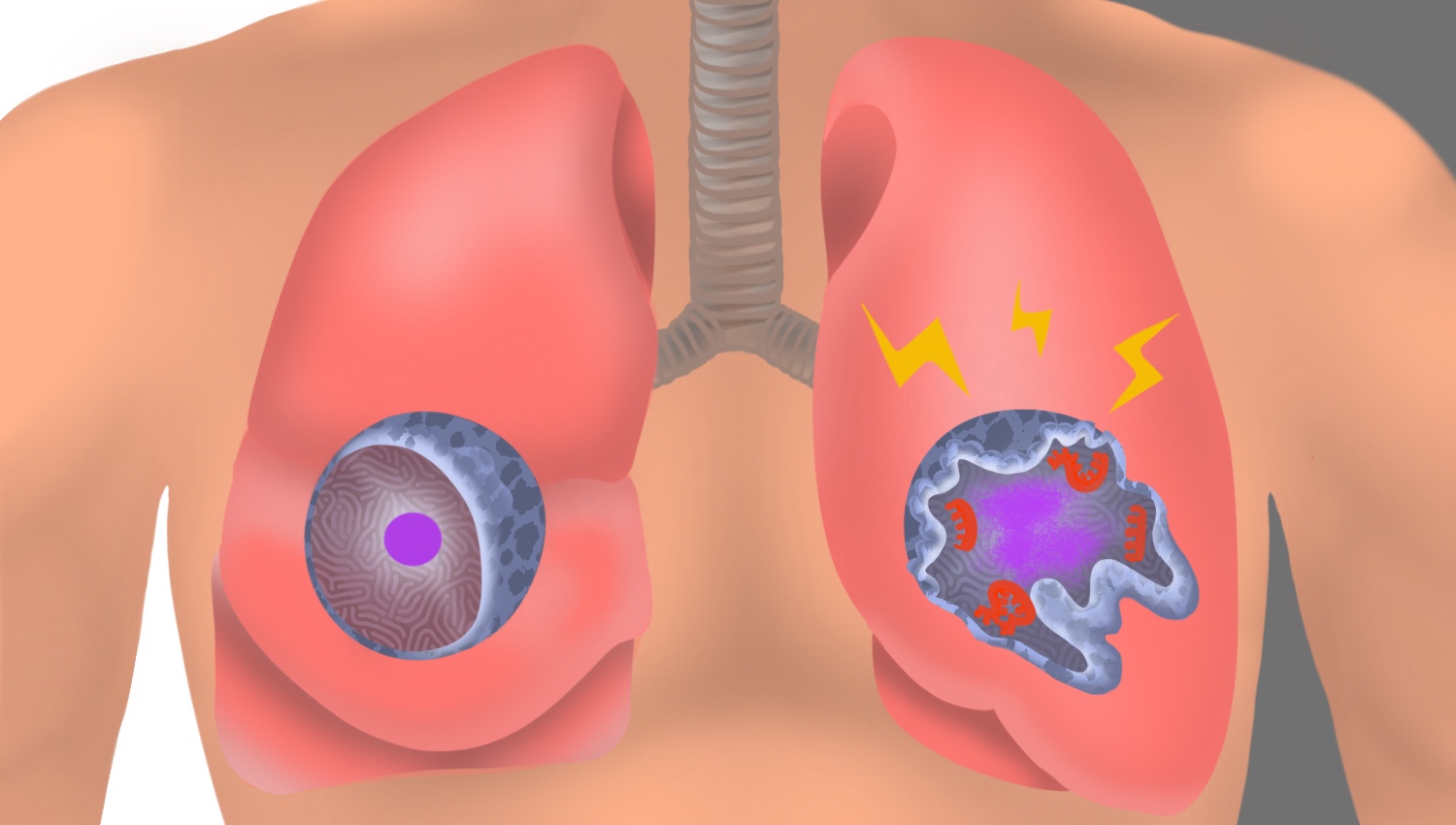
Stanford Medicine study flags unexpected cells in lung as suspected source of severe COVID
A previously overlooked type of immune cell allows SARS-CoV-2 to proliferate, Stanford Medicine scientists have found. The discovery has important implications for preventing severe COVID-19.
Tr1X Announces FDA Clearance of First Investigational New Drug Application for TRX103, an Allogeneic Regulatory T-Cell Therapy to Treat Autoimmune Diseases - Pipelinereview
SAN DIEGO, CA, USA I April 10, 2024 I Tr1X, Inc., an autoimmune and inflammatory disease cell therapy company focused on the development of novel
Collaborative research provides clues to immunity, longer life - Bitterroot Star
by Michael Howell Feeling old and especially vulnerable in the face of the many variations of Flu and RSVP viruses going around? There is good reason to be concerned. Recent […]
Multiple Sclerosis and Epstein-Barr Virus: What Do We Know?
Research suggested that EBV is the primary cause of MS. What are the clinical implications, and could this transform treatment and prevention of this neurodegenerative condition?
Old immune systems revitalized in Stanford Medicine mouse study, improving vaccine response
Those with aging immune systems struggle to fight off novel viruses and respond weakly to vaccination. Stanford Medicine researchers were able to revitalize the immune system in mice.
The Power Of Fitness Wearables with Stanford’s Dr. Michael Snyder
On this episode of FYI, hosts Nemo Marjanovic and Charles Roberts speak with Professor Dr. Michael Snyder, a leading figure in genomics and personalized medicine.
Remission Holds Fast After Five Relapses for Young Woman With Leukemia
None of the treatments designed to fight Camille's acute lymphocytic leukemia worked, until an innovative stem cell transplant at Stanford.
Your microbiome contains trillions of bacteria and is unique like your fingerprint
Trillions of bacteria inhabiting our bodies -- collectively known as the microbiome -- are as unique to an individual as their fingerprint.
The Resilience of Monoclonal Antibodies and their Makers
The road to developing monoclonal antibodies for effectively targeting cancer was paved with tenacity, passion, and strokes of luck.
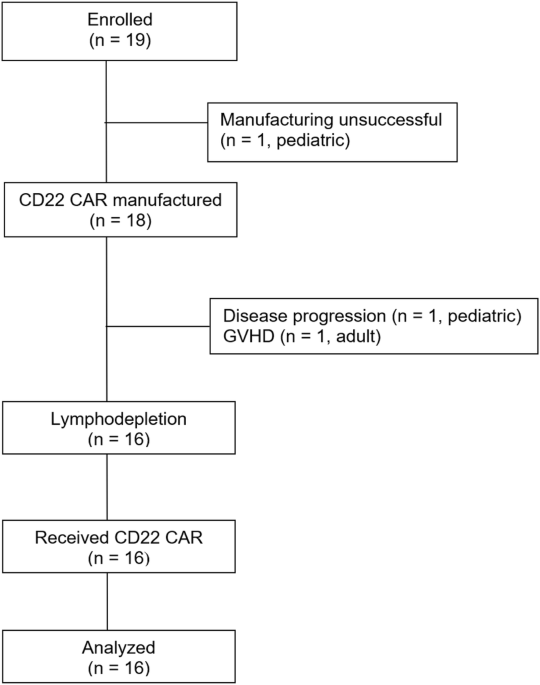
CD22 CAR T cells demonstrate high response rates and safety in pediatric and adult B-ALL: Phase 1b results - Leukemia
Leukemia - CD22 CAR T cells demonstrate high response rates and safety in pediatric and adult B-ALL: Phase 1b results
Remission Holds Fast After Five Relapses for Young Woman With Leukemia
None of the treatments designed to fight Camille's acute lymphocytic leukemia worked, until an innovative stem cell transplant at Stanford.
This protein pic could help develop new cancer treatments
A molecular “snapshot” of a protein can be critical to understanding its function. Scientists at Stanford and NYU have published and investigated a new structure of the protein LAG-3 which could enable the development of new cancer treatments.
Unconventional Paths: How she flipped traditional genomics analysis on its head
Statistics expert Julia Salzman returned to biology and has married her two areas of expertise to design a new form of genomics analysis.
Going beyond B cells in the search for a more multi-targeted vaccine
The ultimate goal: a vaccine with coverage so broad it can protect against viruses never before encountered.
Searching for vaccine variability in the land of the flu
The ultimate goal: a vaccine with coverage so broad it can protect against viruses never before encountered.
The hunt for a vaccine that fends off not just a single viral strain, but a multitude
Stanford Medicine researchers are designing vaccines that might protect people from not merely individual viral strains but broad ranges of them. The ultimate goal: a vaccine with coverage so broad it can protect against viruses never before encountered.
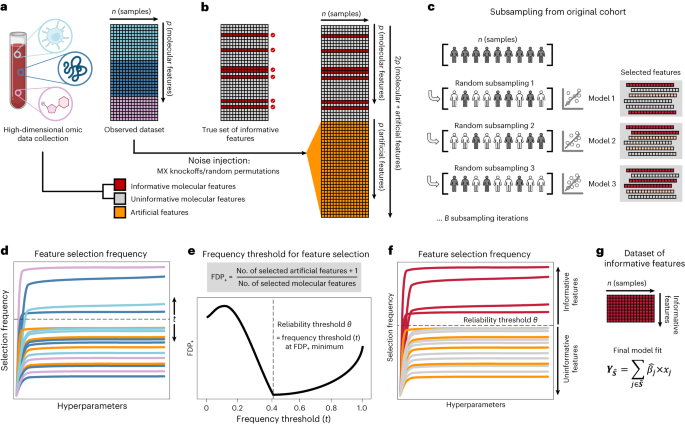
Discovery of sparse, reliable omic biomarkers with Stabl
Stabl selects sparse and reliable biomarker candidates from predictive models.
2023
- – News Center
Engineered human heart tissue shows Stanford Medicine researchers the mechanics of tachycardia
Researchers engineered stem cell-derived heart tissues to study how tachycardia affects the heart and to uncover the inner workings of our body’s engine.
- – News Center
Hodgkin lymphoma prognosis, biology tracked with circulating tumor DNA
Circulating tumor DNA predicts recurrence and splits disease into two subgroups in Stanford Medicine-led study of Hodgkin lymphoma. New drug targets or changes in treatments may reduce toxicity.
- – Stanford Medicine Magazine
Advancing drug development with “petri dish” clinical trials
Testing drugs “in a dish” using heart and blood vessel cells can speed drug development and reduce health care disparities, cardiologist says.
- – News Center
Research findings could explain why young kids rarely get very sick from COVID-19
Children’s noses pack a punch that could help explain COVID-19’s typically mild course in young kids. Researchers hope to parlay that ‘nasal magic’ into increased protections for adults.
- – News Center
Wearable device data reveals that reduced sleep and activity in pregnancy is linked to premature birth risk
Data from wearables show that deviations from normal sleep and activity in pregnancy are connected to a risk for premature delivery, a Stanford Medicine-led study found.
2022
- – News Center
New blood test to identify infections could reduce global antibiotic overuse
A diagnostic test developed by Stanford Medicine scientists can separate bacterial and viral infections with 90% accuracy, the first to meet standards set by the World Health Organization. The new test is described in a paper published Dec. 20 in Cell Reports Medicine. “Antimicrobial resistance is continuously rising, so there has been a lot of effort to reduce inappropriate antibiotic usage,” said Purvesh Khatri, PhD, associate professor of medicine and biomedical data science, and the senior author of the paper. “Accurately diagnosing whether a patient has a bacterial or viral infection is one of the biggest global health challenges.”
- – News Center
Researchers may have found a new path for halting cancer cell production
After finding long, repetitive sequences in the genomes of seven kinds of cancer, researchers at Stanford Medicine and their colleagues developed a molecule that curbed their production. Although the scientists aren’t sure what role the repetitive sequences play in cancer, they were encouraged that they appeared to have found a way to inhibit the creation of more cancer cells. “The most dramatic result was that you could actually target them and stop cell proliferation,” said Michael Snyder, PhD, professor and chair of the department of genetics.
- – Human Performance Alliance
Speeding up bone healing in menopausal females
lder women heal bone fractures slower than men. Now a team has found that a single, localized delivery of estrogen to a fracture can speed up healing in postmenopausal mice. The findings could have implications for the way fractures in women are treated in the future. “The majority of stem cell research is done on male animals. There’s very little research that has actually been done on females,” said Wu Tsai Alliance member Charles Chan, PhD, an assistant professor of surgery at Stanford University and co-senior author of the paper published Oct. 30 in Nature Communications. “The research is long overdue, especially the question of why women heal differently from men.”
- – Pharmacy Times
Expert: Immune Reconstitution With Orca-T Reveals ‘New Realm of Engineered Donor Grafts’ for Patients With Hematologic Malignancies
Pharmacy Times interviewed Everett Meyer, MD, PhD, medical and scientific director of the Cellular Therapy Facility, Stanford Health Care and Assistant Professor of Medicine, Blood and Marrow Transplantation, Stanford University, on the poster presentation titled “Rapid Immune Reconstitution and Elevated Regulatory T Cell Frequencies in Patients Treated with Orca-T” at the 64th American Society of Hematology (ASH) Annual Meeting and Exhibition in New Orleans, Louisiana.
- – Helio
Stanford researcher receives award for lifetime achievement in hematology
Irving Weissman, MD, will receive the Wallace H. Coulter Award for Lifetime Achievement in Hematology at this year’s ASH Annual Meeting & Exposition. Weissman, director of Stanford Medicine’s Institute for Stem Cell Biology and Regenerative Medicine, has made several important contributions to hematology over the past 5 decades.
2021
- – Scope
Who's on first? Duking out scientific paper authorship order
Determining the order of authors on a scientific paper can be tricky. Unless you're a pair of video gaming graduate students. Recently Stanford researcher Garry Nolan, PhD, tweeted about an unconventional way two researchers in his laboratory who had each contributed equally to a study decided who should be listed first on the print version of the paper.
- – BBC
BBC Radio 5 live - 5 Live Science Podcast, Life in plastic, is it fantastic?
As the government moves to Plan B - what actually happens when you catch the new variant?
- – Stanford Professor Garry Nolan Is Analyzing Anomalous Materials From UFO Crashes
Stanford Professor Garry Nolan Is Analyzing Anomalous Materials From UFO Crashes
A Q&A with one of the foremost scientists studying UAPs, and what he hopes to learn by systematically studying bizarre and difficult-to-explain incidents. Dr. Garry Nolan is a Professor of Pathology at Stanford University. His research ranges from cancer to systems immunology. Dr. Garry Nolan has also spent the last ten years working with a number of individual analyzing materials from alleged Unidentified Aerial Phenomenon.
- – Gizmodo
The Coronavirus Can Infect and Possibly Hide in Fat Cells, Study Finds
The preliminary findings could partially explain why people living with obesity are at higher risk of severe covid-19. “This could well be contributing to severe disease,” senior author, Catherine Blish, an immunologist at the Stanford University School of Medicine, told the New York Times. “We’re seeing the same inflammatory cytokines that I see in the blood of the really sick patients being produced in response to infection of those tissues.”
- – Mail Online
Blood from ultra-fit mice 'could hold the key to staving off dementia'
Injections of blood from young adult mice that are getting lots of exercise benefited the brains of sedentary mice the same age, according to a study by the Stanford School of Medicine in California. 'The discovery could open the door to treatments that, by taming brain inflammation in people who don't get much exercise, lower their risk of neurodegenerative disease or slow its progression,' said Professor Tony Wyss-Coray, of the Stanford School of Medicine in California, which carried out the research.
October 7, 2021 - The Edge
How can technology help you track real time data about your health and wellbeing? And why should you track anyway? In this episode, Michael Snyder, professor of genetics, talks about how tracking can help you gain deeper understanding of what is going on in your body at a physiological level. Often, illnesses begin developing in our bodies quietly, before any symptoms begin to show up. Snyder’s research shows that by tracking on a regular basis, we can pre-empt diseases. Health data collection using wearable tech can help us take a proactive approach toward prevention of disease. And like they say, prevention is better than cure.
- – Scope
What to do (and not do) when you win the Nobel Prize
Three of Stanford Medicine's Nobel laureates, including Andrew Fire, George D. Smith Professor of Molecular and Genetic Medicine and Professor of Pathology and of Genetics, offer advice to future winners about hearing the news and what to expect next in their careers.
- – The Scientist Magazine
When the Immune Response Makes COVID-19 Worse
If the immune system makes mistake--reacting late or getting the target wrong--it can amplify the damage wrought by SARS-CoV-2.
- – News Center
Statins may be effective treatment for patients with ulcerative colitis
People with ulcerative colitis who are also taking statins have about a 50% decreased risk of colectomies and hospitalization, according to a Stanford Medicine study. Purvesh Khatri, PhD, associate professor of medicine and of biomedical data science, and his team tracked down a connection between a handful of drugs and decreased symptoms of ulcerative colitis.
- – Business Insider
Why Stanford is spending millions to incorporate Apple Watches and Fitbits into medical care
Stanford is exploring consumer wearable devices, including Apple Watches and Fitbits, to monitor heart rates and predict COVID-19 symptoms. Michael Snyder, professor of genetics, leads many of the health systems' wearable projects and says it's up to health systems to quickly figure out whether the data's useful to doctors and how to efficiently extract it from the devices.
- – Scope
Blood test predicts chances of lymphoma relapse after therapy
Stanford Medicine Scientists have devised a blood test to predict some cancer relapses after patients have already been treated.To understand whether ctDNA tracking might hint at relapse, Miklos, who heads Stanford Medicine's Blood and Marrow Transplantation and Cellular Therapy division, Matthew Frank MD, PhD assistant professor of medicine, and their labs enrolled 72 patients with large B-cell lymphoma, who had received CAR-T cell therapy, which involves genetically engineering certain immune cells to find and eliminate specific cancer cells.
June 23, 2021 – NBC Bay Area
NBC Bay Area: COVID-19 and brain inflammation
Stanford researchers have found signs of inflammation, genetic changes and impaired circuitry in the brains of people killed by COVID-19, important clues to the mysterious “brain fog” and mental struggles reported by many patients. Tony Wyss-Coray, professor of neurology and neurological sciences at Stanford Medicine provides comments.
- – MarketWatch
How COVID vaccine's mRNA technology could help cure other diseases
Scientists and companies are trying to harness the mRNA technology to develop vaccines against cancer and other diseases. Bali Pulendran, Violetta L. Horton Professor And Professor Of Microbiology And Immunology and of Pathology, comments on the future on vaccines.
- – News Center
Stanford researchers find signs of inflammation in brains of people who died of COVID-19
A detailed molecular analysis of tissue from the brains of individuals who died of COVID-19 reveals extensive signs of inflammation and neurodegeneration, but no sign of the virus that causes the disease. Tony Wyss-Coray, PhD, professor of neurology and neurological sciences at Stanford, shares senior authorship with Andreas Keller, PhD, chair of clinical bioinformatics at Saarland University.
- – News Center
Climate change linked to longer allergy season in Bay Area, Stanford study finds
Air levels of pollen and mold spores in the San Francisco Bay Area are elevated for about two more months per year than in past decades, and higher temperatures are to blame, a Stanford Medicine study has found, led by senior author, Kari Nadeau, MD, PhD, professor of medicine and of pediatrics at Stanford School of Medicine.
- – Stanford Today
Faculty Women’s Forum announces 2021 award winners
The 2021 Faculty Women’s Forum Awards honor individuals for their outstanding work supporting women at Stanford through role modeling, allyship, leadership and sponsorship. Stanford Immunology faculty Dr. Joy Wu, an associate professor of medicine (endocrinology, gerontology and metabolism) in the School of Medicine is honored in the Allyship Award category.
- – News Center
Smartwatch data can predict blood test results, study reports
Stanford researchers found that data from smartwatches can flag early signs of some health conditions and predict the results of simple blood tests. Scientists from the lab of Michael Snyder, PhD, professor and chair of genetics, tracked data from smartwatches, blood tests and other tests conducted in a doctor’s office in a small group of study participants.
2020
- – PLOS ONE
Biosciences Proposal Bootcamp: Structured peer and faculty feedback improves trainees’ proposals and grantsmanship self-efficacy
Grant writing is an essential skill to develop for academic and other career success but providing individual feedback to large numbers of trainees is challenging. In 2014, we launched the Stanford Biosciences Grant Writing Academy to support graduate students and postdocs in writing research proposals. Overall, this structured program provided opportunities for feedback from multiple peer and faculty reviewers, increased the participants’ confidence in developing and submitting research proposals, while accommodating a large number of participants.
- – News Center
COVID-19 severity affected by proportion of antibodies targeting crucial viral protein, study finds
A comprehensive study of immune responses to SARS-CoV-2 associates mild disease with comparatively high levels of antibodies that target the viral spike protein. But all antibodies wane within months.
- – Scope
After George Floyd's death, a Black neurosurgeon discusses racism, despair and hope
For Black neurosurgeon Samuel Cheshier, George Floyd's killing confirmed that his country is racist; but the aftermath brought hope that change is possible.
- – BioSci Careers
BioSci Connect: Want to get career advice from alumni mentors? One click connects Biosciences trainees & postdocs with like-minded graduates
Have you ever wanted to know more about careers, but were afraid to ask? Don’t know where to start, or whom to ask? Problem solved.
- – Apple Podcasts
Should This Exist?: Young blood / old brains on Apple Podcasts
This podcast featured Tony Wyss-Coray, the D.H. Chen Professor II and a professor of neurology and neurological sciences, who discovered that proteins found in the blood of young mice may reverse the effects of aging when transfused into older mice.
2019
- – News Center
Stanford scientists reliably predict people’s age by measuring proteins in blood
Protein levels in people’s blood can predict their age, a Stanford study has found. The study also found that aging isn’t a smoothly continuous process.
- – News Center
Stanford researchers program cancer-fighting cells to resist exhaustion, attack solid tumors in mice
CAR-T cells are remarkably effective against blood cancers, but their effect can be transient as the cells become exhausted. Stanford researchers found a way to keep the cells effective in mice with human tumors.
- – News Center
Three professors elected to National Academy of Medicine
Hongjie Dai, Julie Parsonnet and Joseph Wu are among the 90 regular members and 10 international members elected this year to the academy, which aims to provide independent, scientifically informed analysis and recommendations on health issues.
- – BioSci Careers
Devavani Chatterjea discovers link to chronic pain ailment in women
“Never could I have imagined the work I am doing now – not even ten years ago.” - Devavani Chatterjea, PhD, MPH
- – News Center
Protein decoy stymies lung cancer growth in mice, Stanford-UCSF study finds
Researchers at Stanford and UCSF slowed the spread of a type of nonsmall cell lung cancer in mice by neutralizing a single protein that would otherwise set off a chain reaction, causing runaway tumor growth.
2018
- – News Center
Four faculty members appointed to endowed professorships
Andra Blomkalns, Gerald Grant, David Kingsley and Crystal Mackall have been appointed to endowed professorships.
- – News Center
Rosenkranz Prize winner hopes to develop malaria vaccine for pregnant women
Prasanna Jagannathan said the $100,000 prize will allow his lab team to ramp up their research in Uganda.
- – Bay Area Lyme Foundation
Michal Caspi Tal, PhD | Bay Area Lyme Foundation
Michal Caspi Tal, PhD Postdoctoral Research Fellow, Stem Cell Biology and Regenerative Medicine, Stanford University, leads the infectious disease team within Irving Weissman’s lab at Stanford University receives the 2018 Emerging Leader Award ($100,000 grant).
2017
- – News Center
David Schneider appointed chair of microbiology and immunology
David Schneider, whose research focuses on resilience to infection and developing mathematical models to predict recovery and well-being, succeeds Peter Sarnow in post.
- – News Center
Trial led by Mark Genovese wins Clinical Research Forum award
In the trial, a new drug proved safe and effective for hard-to-treat rheumatoid arthritis patients. A national organization of senior researchers named the trial one of the top 10 for 2016.
- – News Center
Fibrosis reversed when ‘don’t eat me’ signal blocked
A common signaling pathway unites diverse fibrotic diseases in humans, Stanford researchers have found. An antibody called anti-CD47, which is being tested as an anti-cancer agent, reverses fibrosis in mice.
- – News Center
Drug combination defeats dengue, Ebola in mice
To develop a potential antiviral treatment, Stanford researchers adopted an unusual approach: Rather than trying to disable viral enzymes, they targeted proteins the infected individual makes — and the virus needs.
- – News Center
15 School of Medicine researchers named CZ Biohub investigators
The researchers will be given funding by the Chan Zuckerberg Biohub to develop tools and technologies that support the organization’s goal of curing, preventing or managing every disease.
2016
- – News Center
Gene activity predicts progression of autoimmune disease
Stanford researchers and their collaborators have found a way to tell whether patients with systemic sclerosis were improving during drug treatment a year before a standard clinical test could.
- – News Center
Samuel Strober awarded $6.6 million from state stem cell agency
The California Institute for Regenerative Medicine awarded Samuel Strober, MD, $6.6 million to study a “deceptively simple” way to help kidney transplant recipients tolerate their new organ.
- – News Center
NIH awards $26.4 million to Stanford researchers for physical activity study
The medical school professors were awarded the grants as part of a large-scale National Institutes of Health program to study the biology of how physical activity improves health.
- – News Center
Three faculty elected fellows of AAAS
Stanford faculty members in medicine and in Earth science have been elected fellows of the American Association for the Advancement of Science.
- – News Center
$25 million awarded to center for study of regulatory science
The FDA is funding a collaboration between Stanford and UCSF to improve the regulatory infrastructure that helps to shape modern biomedical research.